PA Convicted of Fraud for Amniotic Fluid Shot Scheme
A Texas doctor assistant has been convicted of fraud for giving sufferers unapproved injections of amniotic fluid for purported danger support, and then illegally billing Medicare for the footage.
After a 5-day trial, a jury deliberated for only an hour to convict Ray Anthony Shoulders, PA, who had submitted $788,000 in faux claims and took in extra than $614,000 from Medicare for the footage, which aren’t permitted to contend with danger.
The 36-year-feeble, who worked out of a Fort Worth danger management sanatorium, faces as a lot as 240 years in federal jail, according to federal prosecutors.
“Now not fully did this defendant are trying to scam Medicare out of hundreds of thousands of bucks, he did something some distance worse by doubtlessly endangering his sufferers’ health in recommending that they be injected with a drug that had no longer been permitted for that cause,” Leigha Simonton, the U.S. Attorney for the Northern District of Texas, talked about in a observation.
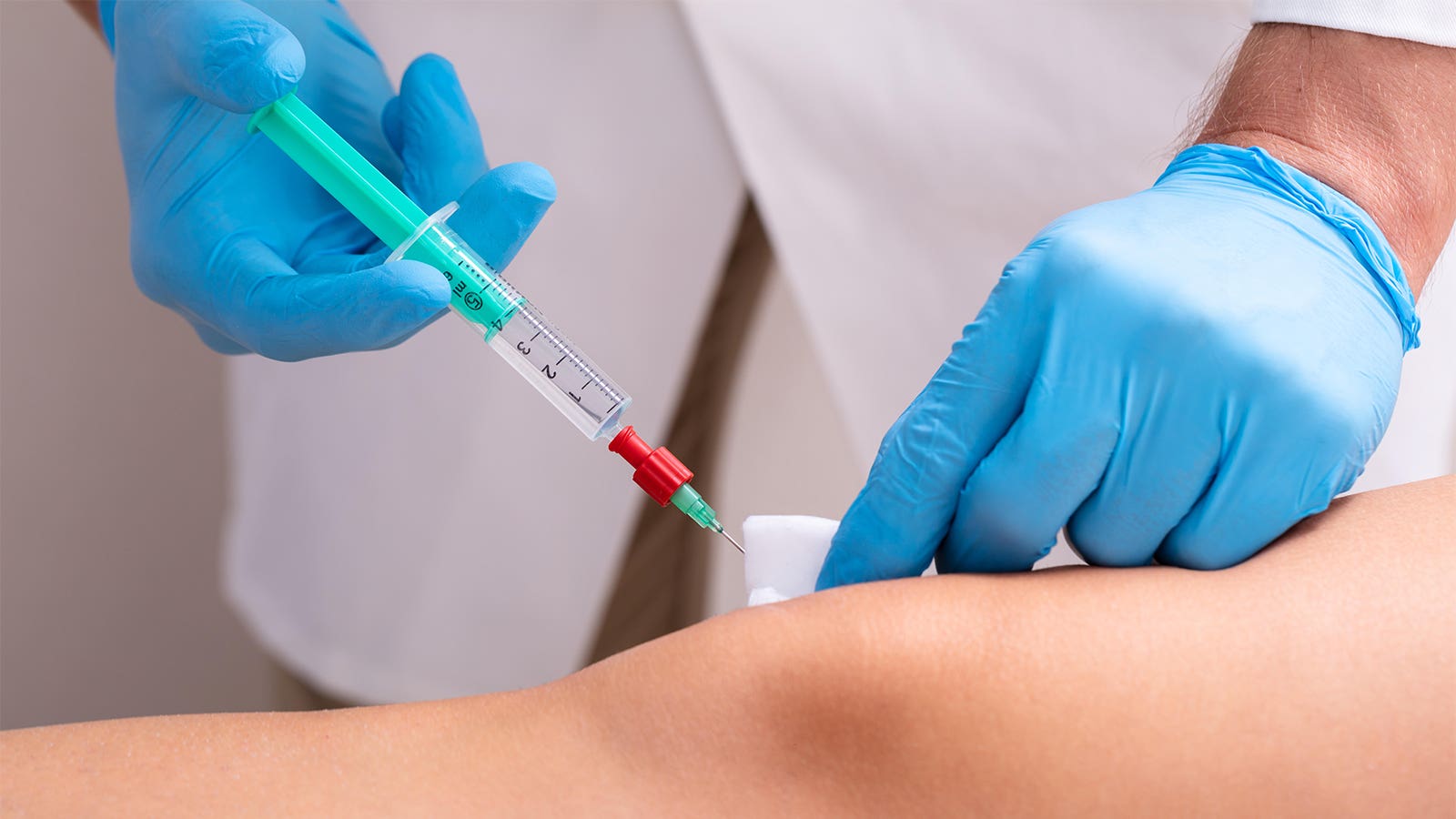
Shoulders gave sufferers footage of an amniotic fluid product known as Cell Genuity, which will not be any longer permitted by the FDA for any indication, federal prosecutors talked about.
Originally, some sufferers paid extra than $800 out of pocket for every shot, however many declined thanks to the high imprint mark, prosecutors talked about.
Then, Shoulders related with a sales earn for but every other amniotic product known as Fluid Circulation, which had a genuine code for compensation, known as a “Q code.”
While Shoulders by no draw bought any Fluid Circulation — it used to be some distance extra expensive than Cell Genuity — he within the kill extinct its genuine compensation code, Q4206, to invoice Medicare for Cell Genuity footage. More sufferers took the footage on legend of they had been lined by insurance coverage, prosecutors talked about.
Shoulders made $1,200 per cc of Cell Genuity; he would fully possess earned about $400 per cc for Fluid Circulation, prosecutors talked about.
From August to October 2020, Shoulders’ sanatorium submitted extra than 100 claims for Fluid Circulation to Medicare, and got about $400,000 in compensation. By November 2020, Shoulders halted the procedure after he turned into cautious that the surprising develop in billing quantity may maybe arrangement off an investigation.
When he noticed no repercussions after 10 months, he started up but again in October 2021 and persisted through December of that year, prosecutors talked about.
Linked to the Shoulders case, in February 2022, a complete lot of U.S. regional Medicare Administrative Contractors introduced that they’d leer to recover losses from “birth tissue” claims going again extra than 2 years. This utilized to amniotic, placental, and umbilical wire injectables, MedPage Currently previously reported.
That legend valuable that some corporations had been in a collection apart to stable Q codes from CMS, which had been in flip marketed to doctors as being reimbursable. It particularly talked about Fluid Circulation by BioLab Sciences the exhaust of code Q4206, and a supply on the second instructed MedPage Currently that the DOJ would doubtless be getting interested, opening the door to prison prosecutions.
Chris Centeno, MD, a health care provider who manages a network of interventional orthopedic clinics, known as Regenexx, and who previously tussled with FDA over stem cell treatments, has long criticized birth-tissue product corporations that promote their products for unapproved orthopedic indications.
“Right here’s unprejudiced correct the tip of a billion-buck claims iceberg as there are hundreds of U.S. clinics which possess fraudulently billed Medicare for these birth tissue products to contend with danger,” Centeno instructed MedPage Currently in an electronic mail. “Most of these clinics are getting their payments clawed again, however I am definite some will pause up losing at trial as smartly.”
In June 2021, the FDA warned that many so-known as regenerative medicines therapies are unapproved, however had been being marketed on the opposite hand to contend with a large kind of ailments and prerequisites. Amniotic fluid — amongst various birth tissue products, equivalent to umbilical wire blood, wire blood stem cells, and Wharton’s jelly — had been on that listing. Utterly different products included stem cells, stromal vascular fraction (additionally known as fats-derived cells), orthobiologics, and exosomes.
The FDA has long been making an strive to set apart a lid on these unapproved cell therapies. In 2017, the company issued guidance on regenerative medicines products, with a November 2020 chop-off date for beefy compliance. That chop-off date used to be extended attributable to the pandemic and went into enact in June 2021. Now, producers must meet FDA’s premarket approval or IND requirements for definite products sooner than selling them to sufferers.
As well, corporations can’t market these products for uses outdoor of the IND, as FDA valuable in a soundless warning letter to exosome maker Kimera Labs.
-
![author['full_name']](data:image/png;base64,R0lGODlhAQABAAD/ACwAAAAAAQABAAACADs=)
Kristina Fiore leads MedPage’s enterprise & investigative reporting team. She’s been a clinical journalist for added than a decade and her work has been identified by Barlett & Steele, AHCJ, SABEW, and others. Ship legend guidelines to okay.fiore@medpagetoday.com. Note
![author['full_name']](https://clf1.medpagetoday.com/media/images/author/kristinaFiore_188.jpg)



